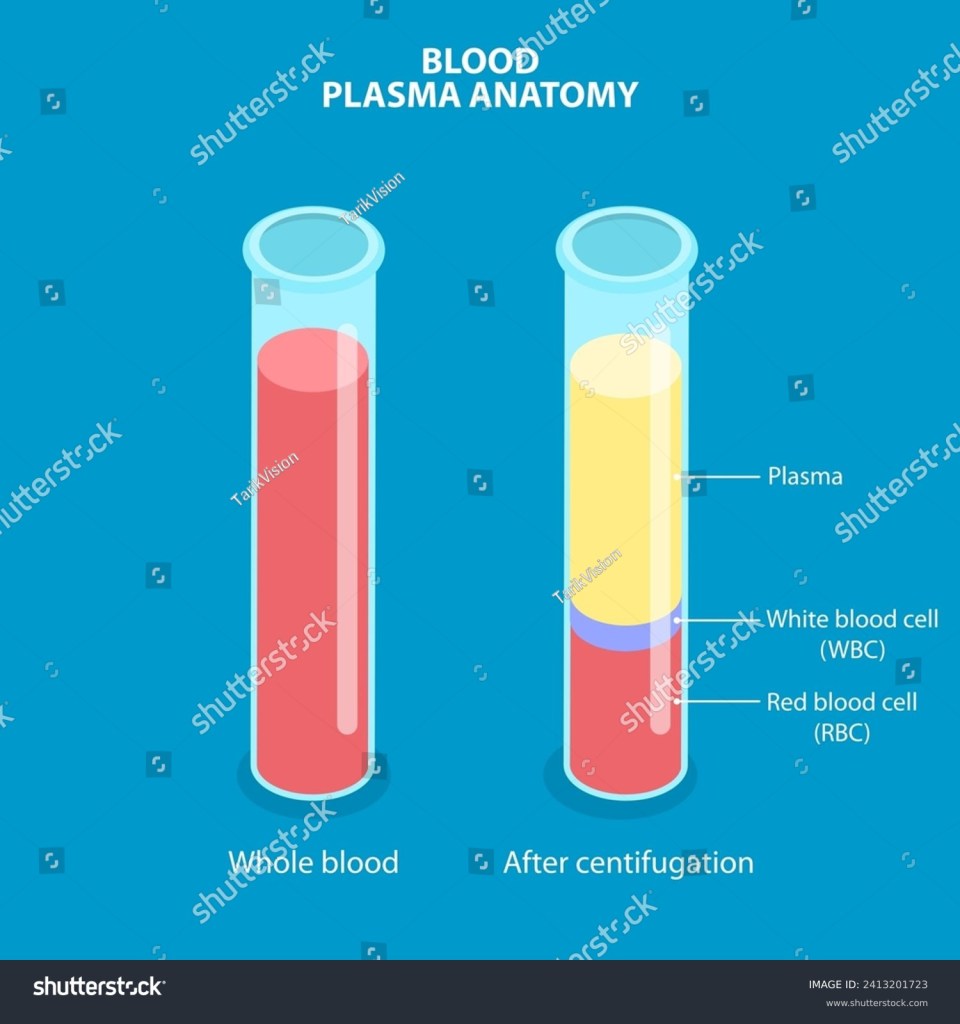
In 2026, publications from The Economics Times to National Geographic, have reported on the unexpected benefits of chewing gum. The U.S. government has a long-released study that confirms this. Nearly 30 years ago, the NIH published a study that found, “[Chewing] increased regional cerebral blood flow in the primary sensorimotor areas by 25-28%, in the supplementary motor areas and insulae by 9-17%, and in the cerebellum and striatum by 8-11%.” (Cerebral blood flow activates the hippocampus, critical for memory, and reduces stress hormones by 16%.) Multiple trials confirm improvements in alertness, sustained attention, and reaction times from chewing gum.
These benefits seem to have been almost forgotten though, as what has been more widely reported in the past decade is the harm that chewing gum causes the body. A study indexed in the NIH database found that chewing commercially available gum can release over 250,000 microplastic particles per hour. Research shows microplastics cross the blood-brain barrier, potentially contributing to oxidative stress and neuroinflammation linked to cognitive decline.
Pertaining to brain injury patients, medical professionals have long known chewing gum may be a boon. The aforementioned statistics note gum’s ability to enhance blood flow and hippocampal stimulation support neural recovery, while stress reduction aids in healing. Gum chewing is used in speech and oral motor rehabilitation therapy, strengthening muscles while stimulating neural pathways, and improving reaction times. Additionally, during these cognitive rehabilitation exercises, gum still retains its ability to reduce stress levels.
To capture benefits while avoiding microplastics and without artificial sweeteners, consider homemade alternatives using natural chicle or beeswax. Many websites offer such recipes, including: Instructables Utopia CookTilYummy. (I do not known the quality or tastiness of these recipes. If you choose to make chewing gum for its cognitive benefits, I recommend using fresh mint as an ingredient because of its own proven cognitive benefits)